What Is The Chemical Makeup Of Most Ash And Resin
Abstract
The paper presents the results of studies on ashes produced from called-for wood biomass in closed wood-fired furnaces, in private household furnaces (Silesian Province, Poland). Dry sieve analysis and detailed granulometric analysis were performed with the Analysette 22 Micro Tec plus analyser. Content of the basic elements (Al, Si, P, Na, K, Mg, Ca, Fe) and potentially toxic elements (As, Pb, Cd, Zn, Cu, Ni, Cr, Hg) was adamant by diminutive absorption spectrometry (AAS) and inductively coupled plasma mass spectrometry (ICP-MS). Relative enrichment factors (REFsouth) were calculated for the elements (Pb, Cd, Zn, Cu, Ni, As, Hg, and Cr), and the emissions (E) of Lead, Cd, Hg, and Every bit to the surroundings were estimated. The carbon content (forty.2%–45.8%), H (3.7%–six.ane%), O (46.two%–l.two%), N (0.12%–0.32%), and S (0.11%–0.96%) in forest biomass varies and depends on the woods species and drying period. The content of volatile parts in forest biomass ranges between 69.3 and 81%. Ash content varies between ii.6 and 18.3%. The forest calorific value ranges from 13.6 MJ/kg to 17.4 MJ/kg. Moisture content in the wood biomass ranges from 13.7% (briquette) to 46.7% (fresh birch). Identification of mineral composition and phases yields a high share of calcite, monetite, fairchildite, and quartz in the examined ashes. The combustion of forest biomass in fireplaces results in increased emissions of Pb and Cd to the atmosphere and may be the cause of introducing pollutants to waters and soils during ash storage.
Introduction
Wood is used for heating homes in many countries. The highest per capita consumption of firewood was recorded in Scandinavian and Baltic countries (5–15 GJ/person) and in Europe (2–x GJ/person) (Deniver et al. 2015).
Individual domestic furnaces in open and closed fireplaces mainly generate ash from burning woods and briquettes produced based on hardwood and coniferous wood. Ash from burning woods biomass is widely recognised equally potentially harmless to the environment. In Poland, in 2018, the consumption of biomass from forest in individual fireplaces reached 13.2% (GUS 2019), while in other Eu countries it was about sixteen% (Faraca et al. 2019). Information technology is estimated that pollution emissions from woods combustion in residential buildings account for approximately 45% of PM2.5 dust and may have a significant impact on the air quality in a given region (Martin et al. 2013; Daellenbach et al. 2016; Maasikmets et al. 2016; Corsini et al. 2017; Czech et al., 2018b, a; Trojanowski and Fthenakis 2019).
The Earth Health Organisation (WHO) believes that such emissions are 1 of the major global environmental run a risk factors (Carvalho et al. 2016; Tao et al. 2016; Vincente and Alves, 2018; Padilla-Barrera et al. 2019). In the process of wood biomass combustion, inorganic components can exist distributed between the gaseous product and the solid ash fraction (Thy et al. 2017). Information technology is estimated that nearly 70% of PM2.5 comes from wood burning in fireplaces and 30% from forest boilers (WHO 2015). The contribution of wood burning to air pollution varies and according to WHO (2015) during the heating season, PM2.five emissions range from 30 to 90% and PM10 emissions range from 10 to 81%. In Denmark, PM2.5 emissions from wood combustion are around 67%. The highest share of PM2.5 from forest combustion was found in the wintertime season in Australia (Launceston) with 95%, followed past the Usa (Fairbanks) with values ranging betwixt 60 and fourscore%. In the southern part of Germany, PM2.5 emissions from wood combustion business relationship for around 59%. In Italy, wood burning in urban households causes an increase in particulate matter from 18 to 76%, and in rural areas from forty to 85% (Siergiej and Jędrak 2019). A typical fireplace for seasoned forest emits up to 1350 mg of particulate matter/grand3 and upwardly to 2180 mg of particulate matter/g3 when called-for damp wood, while a fireplace complying with emission standards only emits about xl mg of particulate affair/10003 (Kubica and Kubica 2016).
According to Alves et al. (2011), the emission factor for biomass combustion in residential buildings is 17.three k/kg PM2.5 (Maenhaut et al. 2012; Martin et al. 2013; Cincinelli et al. 2019). Considering the chemic composition of ash and its potential applications, attention should be paid to the content of alkaline and potentially toxic elements (Czop and Kajda-Szcześniak 2010, 2013; Kajda-Szcześniak 2014). According to Vassilev et al. (2012), dried wood contains: 49.v% C, half-dozen.3% H, 44.2% O, 0.04–0.26% North and 0.2–2.3% mineral compounds. Natural biomass is enriched with such elements as: Ca, Cl, H, K, Mg, Mn, O, and P (Vassilev et al. 2010; Michalik and Wilczyńska-Michalik 2012; Mirowski 2016). CaO, SiO2, KiiO provide for the largest share in wood biomass (Vassilev et al. 2010; Ban and Ramli 2011; Garcia et al. 2015). Ashes from forest combustion too contain potentially toxic elements, such equally Equally, Cd, Ba, Cr, Cu, Ni, Pb, and Zn (Cuenca et al. 2013; Vassilev et al. 2014; Berra et al. 2015; Uliasz-Bocheńczyk and Mokrzycki 2018). According to Jukić et al. (2017) ash from wood called-for is characterised by high Zn and Cu content and depression Cd and Hg content. The phase composition of ashes from biomass mainly includes of calcite, sylvite, arcanite, apatite, anhydrite, periclase, hematite, and unburned carbon (Vassilev et al. 2013; Magdziarz et al. 2018).
The extreme variety of wood biomass burned in households requires a thorough analysis of its physicochemical properties.
This written report aimed to place the potential hazard related to burning wood in fireplace-type furnaces in households. It is of import to determine the concentrations of major, potentially toxic elements and phase composition of ashes every bit this waste material is managed past stored and used in agriculture. About fireplaces in Poland do not run across emission standards and thus contribute to an increase in air pollution with particulate matter. The number of pollutants emitted depends also on the type and quality of wood (e.g. its humidity). We estimated emission to the atmosphere of the most toxic elements (Lead, Cd, Hg, and As), for which data on emission factors tin can be obtained.
Materials and methods
Material for assay
The type of biomass and its physical and chemical parameters are office of the framework quality requirements of a given free energy group. There is a common reference source for quality requirements for solid biomass, which is near frequently the PN-EN ISO 17,225-1:2014-07 standard. Information technology contains specifications for private fuels and classes (PN-EN ISO 17,225-5:2014-07—Part 5: Firewood; PN-EN ISO 17,225-three:2014-07—Part 3: Forest briquettes).
Ash is produced later on burning fresh (birch and alder) and seasoned forest (birch, beech, oak, spruce) as well every bit briquettes made of sawdust from deciduous and coniferous wood (beech and oak—85%, pine chips—15%). Sawdust for the production of briquettes are leftovers from the production of forest accessories. The test samples included ashes from two wood-fired furnaces of the "airtight fireplace" type, each with thermal power of xviii kW and thermal efficiency of approximately 70–72%. The combustion temperature did non exceed 500 °C. Deciduous wood (birch and alder) came from the Andrychów Woods Commune (Małopolskie Voivodeship), beech and oak from the Silesian Voivodeship, and the briquettes were produced in Poland. In each furnace from which the ashes were taken for assay approximately four m3 of wood were burnt during the heating season. We analysed ash from fresh wood (seasoned for about 3 months) and seasoned woods (dried for about 2–3 years). Samples were taken and prepared for analysis in accordance with PN-EN ISO 18,135:2017-06 and PN-EN ISO xiv,780:2017-07 standards.
In full, xiv samples were analysed, 2 samples for each ash type. All the samples of ash from wood combustion with a mass of approximately 1 kg were reduced using the quartering technique (leading to a halving of the sample mass). On completion of this process, we obtained two parts from each ash sample, which were combined and subjected to sieve analysis. For each ash sample, we separated 2 fractions with grain sizes < 100 µm and > 100 µm. For each of the seven ash fractions (except for biomass analyses for C, O, H, Northward, Southward, Cl), nosotros prepared 5 belittling samples. Table i contains the codes for samples of ash from the combustion of wood biomass.
The working methods are presented in the block diagram (Supplementary Data).
Exam method
Control of biomass quality parameters includes assay of ash content, moisture content, volatile parts, and calorific value. The calorific value is an important parameter in biomass assay considering it depends on the moisture and limerick of the burned wood. The moisture content in wood affects its calorific value. The mineral content of wood is determined from the ash content, which depends on the type of woods. The chemical composition of biomass ash features a loftier content of alkali metal compounds which touch its meltability. Ashes from biomass combustion showroom large differences in concentration of potentially toxic elements (e.g. Atomic number 82, Cd, As). The differences in the grain size of the examined ashes result from the blazon of the burned wood. Most often, ashes from wood combustion are characterised by very fine grain size (Mashio et al. 2011).
Dry sieve analysis was performed and ii fractions of ashes were separated for further testing: fine (grain size below 100 µm) and fibroid (grain size in a higher place 100 µm). Analysette 22 Micro Tec plus laser particle size meter was used for detailed granulometric analysis.
Concentrations of basic elements (Na, K, Ca, Mg, Si, Al, Fe) in ash samples for selected wood species were determined using diminutive absorption spectrometry (AAS—Thermo Scientific SOLAAR M6), and for potentially toxic elements (Every bit, Cd, Cu, Cr, Hg, Ni, Lead, Zn, Mn, and Ba), it was determined using ICP-MS technique (mass spectrometry coupled with inductively coupled plasma—Perkin Elmer Sciex Elan 9000).
The ash mineral limerick was determined past X-ray diffraction using model 10'Pert Pro MPD (multi-purpose diffractometer) PW3040/60 X-ray diffractometer of the PANalytical company. Measurement conditions are power supply for the lamp: 40 kV voltage, 40 mA current, analysis range from three° to 75° 2Θ, meter stroke 0.01° Θ and pulse count time—100 s. Estimated mineral content in ash samples was adamant using the Rietveld method. Ash morphology and phase composition were identified with the apply of scanning electron microscope PHILIPS XL 30 equipped with EDS adapter (EDAX Sapphire type).
Unproblematic CHNSO FlashSmart series analyser (Thermo Scientific) was used for analyses of carbon (C), hydrogen (H), nitrogen (N), sulphur (Due south), and oxygen (O) content in tested wood biomass samples. The chlorine content (Cl) was determined using the Variomicro CHNS elementary analyser.
Weight method in accordance with PN-EN ISO 18,122:2016-06 was used to adamant ash content (A). Total moisture content (W) in selected forest species was adamant using the oven-drying method in accord with PN-EN ISO 18,134-2:2017-03. The content of volatile parts (Vdaf) was determined using the weight method in accordance with PN-EN ISO 18,123:2016-01 and the calorific value (Q) was calculated in accordance with PN-EN ISO eighteen,125:2017-07.
Results and discussion
Grain distribution in ashes
What follows from the grain size distribution obtained in sieve ash assay, is that the fraction smaller than 100 µm (min. 67.4% by weight–max. 83.5% past weight) is the largest one, and the fraction higher up 100 µm (min. xvi.5% past weight to max. 32.half dozen% by weight) is smaller (Table ii).
Granulometric analysis using the Analysette 22 Micro Tec analyser yielded a slightly larger percent share of ashes with a fraction lower than 100 µm, at a level of ca. 84% in ashes from seasoned and fresh birch. Ashes from the combustion of wood briquette are characterised by the finest grain size of all analysed wood ash samples. Compared to the other ashes, a lower content of fractions below 100 µm (67.4%) was recorded in the grain composition of ash from the combustion of mixed wood (5A).
Basic biomass components and calorific value
Wood biomass is characterised by very high volatile parts content (min. 69.iii and max. 81%), with a lower value determined for fresh birch (69.iii%) (Table three). Co-ordinate to Vassilev et al. (2010), hateful volatile part content in biomass does not exceed 78%.
Samples of fresh birch (18.3%) and briquette (10.7%) exhibited the highest ash content (Tabular array 3). High ash content in fresh birch is due to high wet content (26.vii%), which affects woods's calorific value (fourteen.9 MJ/kg). The ash content in firewood residues ordinarily ranges betwixt 0.i and xvi.v% (Rybak 2006; Vassilev et.al. 2010; Jagustyn et al. 2011). The wood is normally non available in dry country but exhibits varying humidity levels, which tin can range from 15 to sixty%, depending on how long it has been seasoned in open air. Such a high water content in fuel results from the porosity and hygroscopicity of the wood and its structure (macroporosity) and chemical limerick, mainly the presence of cellulose, hemicellulose, and lignin (microporosity). Especially soft deciduous trees are characterised by about 50% humidity in fresh wood (Bębenek 2008). The moisture content in woods-derived biomass ranges from 13.7 to 46.7%, with the highest moisture content recorded for fresh birch (Table three).
Sulphur and nitrogen constitute undesirable components in biomass due to their harmful touch on the surround. The lowest S content (0.eleven%) was establish in seasoned birch, in other wood biomass samples, the sulphur content varied betwixt 0.71 and 0.96%. Zając et al. (2018) determined sulphur content in ash from the burning of deciduous wood and they found it was in the range from 5631 ppm (birch) to 5107 ppm (oak) whereas in pine ash it was 7142 ppm. Higher values of nitrogen (0.32%) were discovered in wood briquettes when compared to other samples of the tested samples (Tabular array iii).
Table iv presents the content of basic elements in ash. A loftier percentage share of Ca, Yard, P, Al, Si, and Fe was discovered in samples of wood biomass ash. The highest concentrations of these elements were plant in the ash fraction below 100 µm. The tested wood biomass ash samples were characterised by high Fe content ranging from 5.28 to 17.74%. Higher Fe concentrations were found in ash from fresh and seasoned birch and mixed deciduous forest (birch, beech, and oak). Such a loftier Fe content may result from soil contamination in highly urbanised areas (Silesian Voivodeship) from which wood for combustion was sourced. Such high fe content may as well be a result of wood transport and processing. According to various authors (Cuenca et al. 2013; Yeboah et al. 2014; Vassilev et al. 2014), ashes from wood biomass contain 0.09–ane.32% of Fe (Atomic number 262Othree). Higher concentrations of Atomic number 26 (FeiiO3 ranging from 3.9 to 8.89%) in wood biomass ash were determined, among others, by Wang et al. (2008), Koukouzas et al. (2009), and Jukić et al. (2017). Zając et al. (2018) determined Fe content in wood biomass ash at a level of 6518 ppm (birch), 9256 ppm (oak), and 3665 ppm (pino). These authors betoken that the bark of deciduous copse contains lower Fe concentrations (birch—4560 ppm and oak—3841 ppm).
In the tested ash samples of forest biomass, higher concentrations of Ca, Na, Mg, P, Fe, and Ba were establish in fresh birch ash equally compared to seasoned birch ash (Table 4). This mainly concerns ash fraction below 100 µm.
In wood briquette ash, the highest content of volatile parts (81%), Na (5.63%), and P (five.26%) was found in comparison with the other tested ash samples. Jukić et al. (2017), Cuenca et al. (2013), and Koukouzas et al. (2009) determined Na content in wood biomass and plant it to range betwixt 0.16 and 2.05% (Na2O), whereas P content ranged from i.03 to 3.xl% (P2O5).
The highest Thousand content was determined in samples of alder (22.36%), birch, oak, and beech (23.55%) (samples 5A and 5B). The 1000 content in other ash samples tested was in the range between 7.09% (samples 3A and 3B) and 16.66% (samples 6A and 6B). According to Vassilev et al. (2010), the average K2O content in ash from woods biomass and forest combustion is 10.75%. Ban and Ramli (2011) determined K2O content in ash from wood combustion and establish that it was 1.1%. The lowest potassium values were reported by Kowalkowski and Olejarski (2013) in birch ash (0.003%) and bandbox ash (0.03%). Czech et al. (2018b, a) found a high content of alkaline metals (K and Na) in PM2.five emitted from the combustion of birch and softwood pellets. College Na content in the effectively fraction of the examined ashes confirms this hypothesis. When it comes to Thousand content, its share differs slightly depending on ash granulation.
The Mn concentration in the ash samples varies between 0.02 and one.47%. The top concentration of Mn was found in the ash fraction beneath 100 µm. Cuenca et al (2013) and Wang et al (2008) determined Mn content in forest biomass as being in the range 0.ane–0.12%. Co-ordinate to Ciesielczuk et al. (2011), hardwood ash is characterised by high concentrations of Mn (beech—45,193 mg/kg; oak—5462 mg/kg), similarly as pine ash (Mn—308,545 mg/kg) and spruce ash do (7630 mg/kg). Jakubus and Tatuśko (2016) analysed ashes from combustion of wood and institute Mn content of 1088 ppm. Symanowicz et al. (2018) determined Mn content in birch ashes at the level of 919.2 mg/kg, in beech at 840.2 mg/kg, alder at 421.5 mg/kg, and bandbox at 945.7 mg/kg.
The concentration of Ba in the analysed ash samples ranged from 0.03% (samples 5A and 5B) to 1.22% (samples 4A and 4B). According to Lanzertorfer (2015), Ba concentration in ash from mixed leafy and coniferous biomass ranges between 0.009 and 0.048%. Co-ordinate to Uliasz-Bocheńczyk and Mokrzycki (2018), Ba content in birch ash is 0.009% and that of oak ash is 0.001%. Higher Ba content in the tested ash samples may be due to soil contagion in the areas where the trees for combustion were sourced and to precipitation (hard coal combustion in the power industry and domestic furnaces).
The Ti content in wood biomass ash was found to range from 0.22 to 0.37% (Table 4). Higher concentration of this element was found in ash samples from burning briquette and seasoned birch. According to data found in the literature, TiOtwo content in wood ash ranges between 0.35 and 0.78% (Wang et al. 2008; Cuenca et al. 2013; Jukić et al. 2017).
Potentially toxic elements in wood biomass and ashes from its combustion
Concentrations of potentially toxic elements (Pb, Cd, Zn, Ni, Cu, As, Hg and Cr) in the tested ash samples vary profoundly (Table five). The highest Lead contents were institute in ash with a fraction below 100 µm, in samples 3A (1640 mg/kg) and 5A (1198 mg/kg). Szwalec et al. (2016) written report a Pb content of 0.40 mg/kg d.m. in wood briquette. In samples of ash in this fraction (3A, 5A, and 6A), the maximum Zn contents were found, at a level of 1878 mg/kg, 1845 mg/kg, and 1216 mg/kg, respectively (Wiinikka et al. 2013; Szwalec et al. 2016). Such high values are probably due to the combustion of birch with bawl enriched with this element. However, the study by Zając et al. (2018) does not confirm these finding as they found lower Zn content who in birch and oak bark compared to wood coming from these trees. 6A and 7A ash samples showroom higher concentration of As (56–61 mg/kg) compared to the other samples analysed (eleven–38 mg/kg). Higher concentration of Cd was also constitute in 3A (9.half dozen mg/kg) and 7A samples (11.ii mg/kg), whereas samples 1A and 7A (2.1 mg/kg) and 4A (1.eight mg/kg) exhibited high Hg contents. Wisz and Matwiejew (2005) concluded that cadmium content in wood briquette ranged between 0.07 and 0.36 mg/kg.
In the ash fraction in a higher place 100 µm, the content of potentially toxic elements was significantly lower, except for sample 5B where Atomic number 82 was determined at 1112 mg/kg. This is mainly due to the ash granulation, and probably also due to the limerick of the wood briquette, which consists of waste material deciduous wood (beech and oak business relationship for 85%) and coniferous wood (pino chips—15%). The briquette production processes may also matter.
In the tested ash samples, Ni content varied only slightly when ash granulation is taken into account. The highest Ni content was found in samples 2A (76 mg/kg) and 7A. According to Kajda-Szcześniak (2014), the highest Ni content was constitute in ash from the combustion of beech (187.ii mg/kg) and pine (36.98 mg/kg). According to Symanowicz et al. (2018), Ni in ash from wood combustion ranges from 32.8 mg/kg (oak) to 91.three mg/kg (birch). Data from bailiwick literature are characterised by high variability of potentially toxic elements in forest biomass ash (Tabular array five).
Concentrations of potentially toxic elements in biomass samples before combustion can exist written as follows: Zn > Cu > Pb > Cd > Ni > Cr > Hg > As (Table 5). Loftier concentrations of Zn and Pb were plant in samples of biomass from fresh birch and a mixture of deciduous copse (beech, alder, and oak). Zn content in these samples was constitute to range from 55.seven mg/kg to 58.ane mg/kg and Pb concentration from three.62 to 4.xi mg/kg.
The PN-EN ISO 17,225-2-6:2014-07 standard tin can exist used for the assessment of biomass quality with respect to trace elements content. It is a standard adult based on studies of different biomass types conducted in Sweden, Finland, Denmark, the Netherlands, and Federal republic of germany, providing reference values (typical) and occurrence ranges for particular elements. The determined concentrations of potentially toxic elements in wood biomass exceeded those typical of these biomass types establish in the above-mentioned standard.
Relative enrichment factor (REF) in potentially toxic elements for ashes
The enrichment factor allows determining the accumulation of selected elements in ash from wood biomass. This value is characteristic of the specific test area from which the woods biomass samples were taken. The relative enrichment factor (REF) was calculated based on Meij's formula (1994), which takes into account the content of a specific chemical element in the biomass ash (CM ash), the content of this element in biomass (CThousand b) and ash content (A):
$$ {\text{REF }} = {\text{ }}C_{{{\text{ash}}}}^{M} \times {\text{AIC}}_{b}^{M} \times 100. $$
The REF calculation results are presented in Fig. 1.
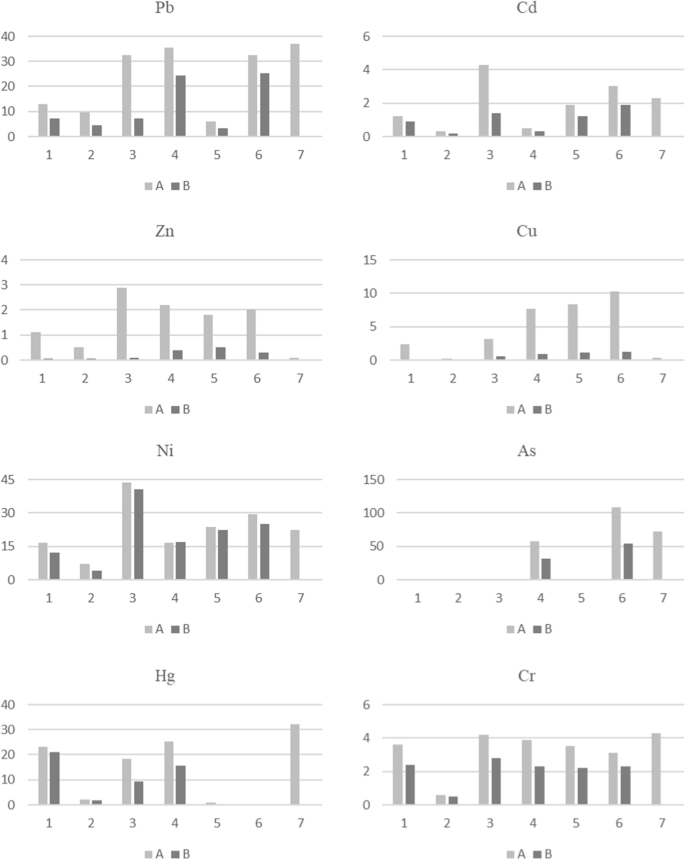
Calculated REFsouthward values in ashes from wood biomass
Ashes from forest biomass combustion are enriched with potentially toxic elements to varying degrees. In woods biomass ash samples, the calculated REFs are high for Pb, Ni, and Hg (Fig. 1). These are ashes from the combustion of fresh birch, mixed deciduous and/or coniferous wood, and wood briquette. In these samples, the REF values are at a similar level. For Ni, the REF values are like in wood biomass ash samples with fractions both below 100 µm and above 100 µm. The highest REF value was calculated for As in ash from burning a mixture of deciduous wood (alder and oak), deciduous and coniferous forest (beech and spruce), and wood briquette. In the remaining forest samples, Equally content was non determined (Tabular array 5). Significantly lower REF values (2–10) were calculated for the remaining elements (Cd, Zn, Cr, and Cu) (Fig. i). Zn and Cu are remarkable as for them REF values are significantly lower in the ash fraction above 100 µm, compared to the ash fraction below 100 µm.
Estimated emissions of Pb, Cd, Hg, and As from burning wood in a closed furnace
Reduction of emissions of potentially toxic elements is one of the fundamental elements of European union ecology policy. Determination of the amount of potentially toxic pollutants emitted to the atmosphere is of corking importance for measures taken to improve air quality (Maenhaut et al. 2012; Martin et al. 2013; Cincinelli et al. 2019). Wood-fired furnaces found in individual households, similarly to hard coal-fired furnaces, are point sources of atmospheric pollution. Households mostly emit them during the winter season.
The corporeality of emissions introduced into the atmosphere depends on the type of fuel, consumption, and fuel parameters. For the calculation of Pb, Cd, Hg, and As emissions from wood combustion, we used the data of the National Centre for Balancing and Direction of Emissions (KOBIZE) in Poland for the yr 2019, adopting the emission factor for wood determined in accordance with the recommendations of the European Environment Agency (EEA Report 2019).
Emission book (E) was calculated per unit of energy (1 GJ) based on the relationship including which wood consumption (B), wood calorific value (West0), and emission cistron (W) of the element in question:
$$ E{\text{ }} = {\text{ }}B \cdot W_{0} \cdot W~~\left[ {{\text{mg}}/{\text{GJ}}} \right]. $$
Table 6 presents the calculated factors of Pb, Cd, Hg, and As emissions to the atmosphere subsequently combustion of forest biomass. For calculations, nosotros assumed 4 miii of burnt wood and four Mg of briquette. The 1 mthree weight for particular wood species, depending on the moisture content, ranged from 530 kg (alder) to 750 kg (birch).
Combustion of wood briquettes resulted in the highest emissions of lead, cadmium, mercury, and arsenic to the atmosphere. It probably results from the blazon of waste that is used for the production of briquettes. In this case, it included very fine sawdust generated in the procedure of production of woods products (deciduous and coniferous wood). Combustion of fresh and seasoned birch also results in higher, compared to other woods species, emissions of Atomic number 82, Cd, Hg, and As to the atmosphere (Tabular array 6). Studies take shown that the lowest emissions of Pb, Cd, Hg, and Every bit to the temper were achieved when called-for a mixture of beech and spruce. However, spruce is not woods species recommended for closed furnaces due to high resin content.
Literature data well-nigh ofttimes refer to the estimated emission of PM10 and PM2.5 to the temper from wood combustion. According to a study by Czech et al. (2018b, a), PM2.v emitted from birch combustion is enriched with Zn and the average emission factor is 0.683 µg/GJ. Research conducted by Schmidl et al. (2007) indicates that emissions from the combustion of pellets and wood fries in manually loaded wood-fired furnaces amount to 0.09 kg/GJ. Kistler et al. (2012) have shown that the highest grit emissions to the atmosphere resulted from the combustion of oak (0.22 kg/GJ). Trojanowski and Fthenakis (2019), in their studies on dust emissions to the atmosphere from forest combustion in residential buildings, showed that the vast majority of the formed particles fall within the nanometre (NP) range. The size of the emitted particles is influenced by the type of wood and the type of furnace (fireplace).
Forest-fired furnaces of the fireplace type that do not comply with emission standards may provide grit emissions in the range of 1350 mg/mthree (dry wood) to 2180 mg/grand3 (wet wood) (Siergiej and Jędrak, 2019). Out of all the PM 2.5 grit from wood combustion, lxx% comes from fireplaces and 30% from fired boilers (WHO 2015). Other authors (Bari et al. 2011; Amaral et al. 2016) also betoken to the high emission of fine particles (< 10 µm) to the atmosphere every bit a result of biomass combustion. The particulate matter emission rate from biomass combustion in residential buildings is 17.3 g/kg PM2.v (Alves et al. 2011).
Wood burning tin cause an increase in particulate matter concentration, which in plow causes heart irritation, affects the respiratory system, can crusade heart rhythm issues, and atomic number 82 to stroke (Riddervold et al. 2012; Robinson 2015; Eze et al. 2015; Morakinyo et al. 2016; Shi et al. 2016; Kasangana et al. 2017).
Phase composition of wood biomass ash
Mineral content in the tested ash samples was adamant by X-ray diffraction. In all tested ash samples with the < 100 µm fraction, dominant components included calcite (CaCO3), fairchildite (Chiliad2Ca(COiii)two), periclase (MgO), monetite (CaHPO4), and quartz (SiOii). The percentage share of these minerals varies depending on the type of woods biomass burnt. Ashes from burning fresh birch (3A) and seasoned birch (2A) contain the most calcite (about 57%) in comparison with other tested ash samples (23%–37%). The estimated content of fairchildite, (19%–23%) and monetite (8%–16%) was higher in samples of ash from the incineration of seasoned birch (2A) and alder (1A) compared to the other ash samples.
In ashes with a fraction higher up 100 µm, the main minerals included calcite and quartz. In fresh (3B) and seasoned (2B) birch ash, calcite content was estimated at 83% and 64%, respectively. Michalik and Wilczyńska-Michalik (2012) found high quartz content in beech bark ash (56%–58%). The share of quartz in these ash samples ranged from 25 (3B) to 32.5% (2B). Auxiliary components in the tested ashes (2B, 3B, 4B, 5B, 6B) included periclase (MgO), apatite(Cafive(PO4)3(OH,F,Cl), sylvite (KCl), dolomite ([CaMg(CO3)ii]), calcium oxide (CaO), and portlandite (Ca(OH)2).
The crystal construction models were calculated using the ICDD database. Data on the quantitative, relative content of crystalline phases (the method does not take into account the amorphous phase) was obtained based on the Rietveld method, which consists of calculating the theoretical diffractogram and fitting information technology, using the to the lowest degree-squares method, to the experimental diffractogram. The amount of the "t" stage after running the Rietveld method was estimated using the following formula (Gąsiński and Poszytek, 2013):
$$ S_{t} = {\text{ }}{{A_{t} B_{t} M_{t} V_{t} } \mathord{\left/ {\vphantom {{A_{t} B_{t} M_{t} V_{t} } {\sum _{due north} A_{n} B_{n} M_{n} V_{n} }}} \right. \kern-\nulldelimiterspace} {\sum _{n} A_{n} B_{north} M_{n} V_{n} }} $$
where
S t —weight share of the crystalline phase s in the i-phase mixture.
A t —phase scale gene due south.
B t —number of formal units in the elementary prison cell of stage s.
Thou t —weight of the formal unit of stage s.
V t —book of the elementary cell of phase s.
n—number of modelled crystalline phases.
The content of these components calculated was estimated using the Rietveld method at less than 4%. Amongst mineral components, calcite (93%) and portlandite (vii%) content were determined in sample 1B (alder ash). As a soil component, quartz is very oft found in ash from woods biomass (Bostrom et al. 2012; Sano et al. 2013). The high content of Al, Thou, and Na in wood biomass ash results from burning intense combustion of fossil fuels in a given area and their concentration in soil and tree bark (Sano et al., 2013). Their phytoavailability is expressed past the post-obit relationship: Thou > Ca > Mg < P.
Ash analyses by means of scanning electron microscopy allowed us to make up one's mind the morphology and chemical limerick of private particles and ash aggregates. The monetite substance in ashes from combustion of wood biomass usually contains admixtures of Mg, K, and Mn in variable amounts (Fig. 2). It coexists with calcite and Mg, Mn, and Ba oxides forming openwork forms and aggregates which mutually overgrow one another. Inclusions of fairchildite and portlandite are also nowadays in this substance.
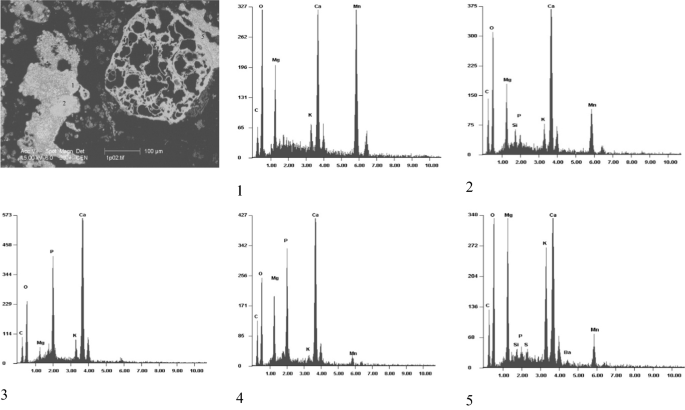
SEM and EDS images of particles in ashes from combustion of birch seasoned at 500 °C
The pores being the residue after wood combustion are usually filled with a substance consisting of iron and titanium oxides (Fe2O3;TiOii) and aluminosilicate substances and aggregates (Fig. 3).
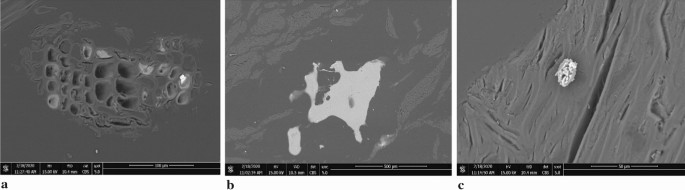
SEM images showing phases in ash from seasoned birch combustion (a—calcite with Mn in the forest structure after called-for; b—quartz; c—calcite with Mn and Ba)
The phase limerick of ash from wood biomass combustion includes inorganic components, both amorphous and crystalline (Vassilev et al. 2013). Many authors (Maschio et al. 2011; Jukić et al. 2017; Maresca et al. 2017) betoken that the master components in ash from wood combustion include quartz, calcite, periclase, and calcium oxide. Maresca et al (2017) also report the presence of hematite (FetwoOiii) in spruce ash.
Calcite, which is present in the fibrous woods tissues, was also observed in woods biomass (Fig. 3a). Quartz mainly fills the cavities between bark crumb and pulp (Fig. 3b). Calcite is most ofttimes plant on the wood surface and contains Mn and/or Ba (Fig. 3b, c). Ashes from the combustion of wood that has a long growth period may accept a different mineral composition and higher concentrations of heavy metals (Nunes et al. 2016).
Statistical analysis of results
Basic statistical parameters were calculated for the test results: coefficient of variation (CV) and variance (5). Using Pearson's linear correlation coefficient, an endeavour was made to determine the relationship between potentially toxic elements and ∑Si + Al and ∑Ca + K (Tabular array 7). The coefficient of variation, like the standard divergence, is used to investigate the degree of variation in the value of a variable. The calculated coefficient of variation in the range of 0–20% for potentially toxic elements is indicative of low variation in the chemic composition of wood ash. The calculated variance values are indicative of high variability of Lead (1A and 1B), Zn (1A, 2A, 4A, 4B, 6A, 6B), and Cu (6A) in relation to the mean value in the tested ash samples. The correlation coefficient Pearson (r = 0.00–0.10 negligible correlation; r = 0.10–0.39 weak correlation; r = 0.forty–0.69 moderate correlation; r = 0.70–0.89 stiff correlation; r = 0.90–i.00 very strong correlation) was used to appraise the human relationship between the determined elements (Schober et al. 2018). Loftier correlation coefficients values (r = 0.70–0.89) were constitute for potentially toxic elements (Pb, Cd, Zn, Cu, Every bit, and Ni) confronting ∑Ca + K. This is mainly due to the loftier content of element of group i components (Ca, K, Na) in natural biomass. The highest Ca content was found in tree bark (Mirowski et al. 2018). Moderate correlation coefficients were found for potentially toxic elements relative to the ∑Si + Al sum. The average content of the most mutual oxides in the chemical composition of natural woody biomass, tin be represented every bit follows (Vassilev et al. 2010): CaO > SiO2 > K2O > MgO > Al2O3 > PtwoO5. The lower concentration of Si and Al in the natural woody biomass and the investigated ash samples affects the correlation with potentially toxic elements. A big number of potent and moderate significant correlation coefficients is indicative of statistically significant relationships that exist between the determined potentially toxic elements and the concentrations of ∑Ca + K and ∑Si + Al.
Further research in this area, on a larger sample, is being carried out, and the results obtained will allow to draw correct conclusions and perform correct statistical calculations.
Conclusion
The blazon of wood, its storage time, and conditions influence the calorific value and thus on the free energy efficiency during combustion and, above all, the chemic composition of the generated ash.
In the ash fraction below 100 µm, high contents of Ca, Thousand, Si, and Fe and potentially toxic elements were recorded. The high concentration of Atomic number 26 in wood biomass ash is probably determined by the mineral limerick of soils and the location of forest harvesting. In the phase composition of seasoned wood, we determined iron oxides, which can be plant in fibrous tissues.
Relative enrichment factors (REFsouthward) calculated for As, Atomic number 82, Ni, and Hg in ashes were significantly higher compared to the other elements (Cd, Cu, Zn, Cr). The results obtained are indicative of the demand to monitor the content of potentially toxic elements in burnt wood biomass, due to the high variability of the concentration of these elements.
The average calculated values of Pb, Cd, Hg, and As emissions for the examined ash are the highest for the combustion of wood briquettes and the lowest for the combustion of spruce. Combustion of these wood species may contribute to the deterioration of air quality in areas where fireplace furnaces are used to heat houses.
The phase composition of ash from wood biomass is dominated by calcite, fairchildite, periclase, monetite, and quartz. Their percentage share depends on the type of woods biomass.
Farther research is needed taking into account the chemical backdrop of wood biomass and its commercial products equally additional assessment criteria.
Conflicts of involvement
The authors declare no disharmonize of interest.
References
-
Alves C, Gonçalves C, Fernandes AP, Tarelho L, Pio C (2011) Fireplace and forest stove fine particle emissions from combustion of western Mediterranean woods types. Atmospheric Res 101:692–700. https://doi.org/ten.1016/j.atmosres.2011.04.015
-
Amaral SS, Carvalho JA Jr, Martins-Costa MA, Pinheiro C (2016) Particulate matter emissions factor for biomass combustion. Temper vii:141. https://doi.org/10.3390/atmos7110141
-
Ban CC, Ramli G (2011) The implementation of wood waste matter ash as a fractional cement replacement textile in the production of structural course concrete and mortar: An overview. Resour Conserv Recycl 55:669–685. https://doi.org/10.1016/j.resconrec.2011.02.002
-
Bari MA, Baumbach G, Kuch B, Scheffknecht C (2011) Air pollution in residential areas from wood-fired heating. Aerosol Air Quality Res 11:749–757. https://doi.org/10.4209/aaqr.2010.09.0079
-
Berra M, Mangialardi T, Paolini AE (2015) Reuse of woody biomass wing as in cement-based materials. Constr Build Mater 76:286–296. https://doi.org/10.1016/j.conbuildmat.2014.11.052
-
Bębenek Z (2008) Biopaliwa stałe [w:] Odnawialne i niekonwencjonalne źródła energii, Gałusza M, Paruch J (red.), Tarbonus, Kraków-Tarnobrzeg (Poland).
-
Bostrom D, Skoklung N, Grimm A, Boman C, Ohman M, Brostrom Thou, Backman R (2012) Ash transformation chemical science during combustion of biomass. Free energy Fuels 26:85–93. https://doi.org/10.1021/ef201205b
-
Carvalho RL, Jensen OM, Luís AC (2016) Mapping the operation of wood-burning stoves by installations worldwide. Energy Build 127:658–679. https://doi.org/10.1016/j.enbuild.2016.06.010
-
Czech H, Miersch T, Orasche J, Abbaszade Chiliad, Sippula O, Tissari J, Michalke B, Schnelle-Kreis J, Streibel T, Jokiniemi J, Zimmermann R (2018a) Chemical limerick and speciation of particulate organic matter from modern residential small-scale wood combustion appliances. Sci Total Environ 612:636–648. https://doi.org/ten.1016/j.scitotenv.2017.08.263
-
Ciesielczuk T, Kusza G, Nemś A (2011) Fertilization with biomass ashes as a source of trace elements for soils. Instytut Ochrony Środowiska—Państwowy Instytut Badawczy, Warszawa, Poland. Ochrona Środowiska i Zasobów Naturalnych, vol 49, pp 219–227. ISSN 1230–7831
-
Cincinelli A, Guerranti C, Martellini T, Scodellini R (2019) Residential wood combustion and its impact on urban air quality in Europe. Current Stance Environ Sci Health 8:10–14. https://doi.org/10.1016/j.coesh.2018.12.007
-
Czop M, Kajda-Szcześniak M (2010) Content of heavy metals in ashes later called-for biomass briquette. Archives Waste matter Manage Environ Protect 12(1):67–76
-
Czop M, Kajda-Szcześniak M (2013) Environment bear upon of straw based fuel combustion. Athenaeum Waste material Manage Environ Protect 39(4):71–80. https://doi.org/ten.2478/aep-2013-0031
-
Corsini Due east, Vecchi R, Marabiin L, Fermo P, Becagli Due south, Bernardoni V, Caruso D, Corbella L, Dell'Acqua M, Galli CL, Lonati G, Ozgen Due south, Papale A, Signorini South, Tardivo R, Valli Chiliad, Marinovich M (2017) The chemical composition of ultrafine particles and associated biological effects at an alpine boondocks impacted by wood burning. Sci Total Environ 587–588:223–231. https://doi.org/10.1016/j.scitotenv.2017.02.125
-
Cuenca J, Rodríguez J, Martín-Morale SM, Sánchez-Roldá Z, Zamorano M (2013) Furnishings of olive residue biomass wing ash every bit filler in cocky compacting physical. Constr Build Mater twoscore:702–709. https://doi.org/10.1016/j.conbuildmat.2012.09.101
-
Czech H, Miersch T, Orasche J, Abbaszade Yard, Sippula O, Tissari J, Michalke B, Schnelle-Kreis J, Streibel T, Jokiniemi J, Zimmermann R (2018b) Chemic composition and speciation of particulate organic thing from modern residential small-scale wood combustion appliances. Sci Total Environ 612:636–648. https://doi.org/ten.1016/j.scitotenv.2017.08.263
-
Daellenbach KR, Bozzetti C, Krepelová A, Canonaco F, Wolf R, Zotter P, Fermo P, Crippa One thousand, Slowik JG, Sosedova Y, Zhang Y, Huang RJ, Poulain L, Szidat S, Baltensperger U, Haddad IE, Prévôt ASH (2016) Characterization and source apportionment of organic aerosol using offline droplets mass spectrometry. Atmos Meas Tech ix:23–39. https://doi.org/10.5194/amt-nine-23-2016
-
Deniver van der Gon HAC, Bergström R, Fountoukis C, Johansson C, Pandis SN, Simpson D, Visschedijk AJH (2015) Particulate emissions from residential wood combustion in Europe—revised estimates and an evolution. Atmos Chem Phys fifteen:6503–6519. https://doi.org/x.5194/acp-fifteen-6503-2015
-
EEA—European Environment Agency Report, (2019) Air quality in Europe—2019 written report
-
Eze IC, Hemkens LG, Bucher HC, Hoffman B, Schindler C, Künzli Due north, Schikowski T, Probst-Hensh NN (2015) Clan between ambient air pollution and diabetes mellitus in Europe and N America: systematic review and meta-analysis. Environ Health Perspect 123(five):381–389. https://doi.org/10.1289/ehp.1307823
-
Faraca 1000, Boldrin A, Astrup T (2019) Resource quality of wood waste: THE importance of physical and chemical impurities in wood waste for recycling. Waste material Manage 87:135–147. https://doi.org/10.1016/j.wasman.2019.02.005
-
Garcia R, Pizarro C, Alvarez A, Lavin AG, Bueno JL (2015) Written report of biomass combustion wastes. Fuel 148:152–159. https://doi.org/x.1016/j.fuel.2015.01.079
-
Gąsiński A, Poszytek A (2013) Zastosowanie metody Rietvelda do ilościowego oznaczania skłądu mineralnego skał łupkowych—implikacje practice poszukiwań i eksploatacji niekonwencjonalnych złóż gazu ziemnego (gazu "łupkowego"). Nafta-Gaz 3:213–229 ((in Smooth))
-
GUS—Statistics Poland—Statistical analyses, (2019) Energy consumption in households in 2018, Warsaw 2019 (Poland)
-
Jagustyn B, Bątorek-Giesa Due north, Wilk B (2011) Ocena włąściwości biomasy wykorzystywanej do celów energetycznych. CHEMIK 65(half dozen):557–563
-
Jakubus One thousand, Tatuśko N (2016) Evaluation of metals bioavailability in agronomical soil treated with wood biomass. Polish J Soil Sci 2:195–297. https://doi.org/ten.17951/pjss/2016.49.2.195
-
Jukić M, Ćurković L, Šabarić J, Kerolli-Mustafa Thousand (2017) Fractionation of heavy metals in fly ash from wood biomass using the BCR sequential extraction procedure. Bull Environ Contam Toxicol 99:524–529. https://doi.org/10.1007/s00128-017-2160-0
-
Kajda-Szcześniak M (2014) Characteristic of ashes from fireplace. Archives Waste material Manage Environ Protect xvi(three):73–78
-
Kasangana KK, Chadyiwa M, Masekameni D, Makonese T (2017) Exposure to forest dust and wellness furnishings: a review of epidemiological evidences from developing countries. In: Conference: National Association for Make clean Air At: Johannesburg, South Africa
-
Kistler Chiliad, Schmidl C, Padouvas East, Giebl H, Lohninger J, Ellinger R, Bauer H, Puxbaum H (2012) Odor, gaseous and PM10 emissions from small calibration combustion of forest types indigenous to Fundamental Europe. Atmos Environ 51:86–93. https://doi.org/10.1016/j.atmosenv.2012.01.044
-
Koukouzas N, Wardb CR, Papanikolaoua D, Li Z, Ketikidis C (2009) Quantitative evaluation of minerals in fly ashes of biomass, coal and biomass-coal mixture derived from circulating fluidised bed combustion technology. J Hazard Mater 169:100–107. https://doi.org/10.1016/j.jhazmat.2009.03.116
-
Kowalkowski A, Olejarski J (2013) Possibilities of using ashes from forest biomass every bit a source of nutrients (Możliwości wykorzystania popiołów z biomasy leśnej jako źródła elementów odżywczych). In: Biomasa leśna na cele energetyczne. Carmine. naukowa P. Gołas i A. Kaliszewski. Prace Instytutu Badawczego Leśnictwa, Sękocin Stary, 147−176 (in Shine)
-
Kubica K, Kubica R, (2016) Założenia bazy danych wskaźników emisji dla kalkulatora emisji zanieczyszczeń z urządzeń grzewczych na paliwa stałe. Raport dla Instytutu Ekonomii Środowiska, Gliwice, pp 1–18 (in Shine)
-
Lanzerstorfer C (2015) Chemical composition and physical properties of filter wing ashes from eight grate-fired biomass combustion plants. J Environ Sci 30:191–197. https://doi.org/10.1016/j.jes.2014.08.021
-
Maasikmets M, Kupri HL, Teinemaa East, Vainumäe Yard, Arumäe T, Roots O, Kimmel 5 (2016) Emissions from burning municipal solid waste product and forest in domestic heaters. Atmospheric Pollut Res seven:438–446. https://doi.org/ten.1016/j.apr.2015.x.021
-
Maenhaut Westward, Vermeylen R, Claeys One thousand, Vercauteren J, Matheeussen C, Roekens East (2012) Assessment of the contribution from wood burning to the PM10 aerosol in Flanders, Belgium. Sci Total Environ 437:226–236. https://doi.org/10.1016/j.scitotenv.2012.08.015
-
Magdziarz A, Gajek Thou, Nowak-Woźny D, Wilk M (2018) Mineral phase transformation of biomass ashes—Experimental and thermochemical calculation. Renew Energy 128:446–459. https://doi.org/10.1016/j.renene.2017.05.057
-
Maresca A, Hyks J, Astrup TF (2017) Recirculation of biomass ashes onto wood soils: ash composition, mineralogy and leaching proprieties. Waste material Manage 70:127–138. https://doi.org/10.1016/j.wasman.2017.09.008
-
Martin 1000, Tritscher T, Jurányi Z, Heringa MF, Sierau B, Weingartner Eastward, Gysel K, Prévôt ASH, Baltensperger U, Lohmann U (2013) Hygroscopic backdrop of fresh and anile wood burning particles. J Aerosol Sci 56:fifteen–29. https://doi.org/10.1016/j.jaerosci.2012.08.006
-
Maschio S, Tonello Thousand, Piani L, Furlan E (2011) Fly and bottom ashes from biomass combustion equally cement replacing components in mortars production: Rheological behaviour of the pastes and materials compression strength. Chemosphere 85:666–671. https://doi.org/10.1016/j.chemosphere.2011.06.070
-
Meij R (1994) Trace element behavior in coal-fired power plants. Fuel Process Technol 39:199–217
-
Michalik Yard, Wilczyńska-Michalik W (2012) Mineral and chemical composition of biomass ash. Europ Mineralogical Briefing 1:423. https://doi.org/10.13140/2.1.4298.5603
-
Mirowski T (2016) Utilization of biomass for energy purpose versus reduction of emission of air pollutants from municipal and household sector. Rocznik Ochrony Środowiska xviii:466–477
-
Mirowski T, Mokrzycki E, Uliasz-Biocheńczyk A (2018) Energetyczne wykorzystanie biomasy. Wyd. IGSMiE PAN, Kraków (in Shine)
-
Morakinyo OM, Mokgobu MI, Mukhola MS, Hunter RP (2016) Heath outcomes of exposure to biological and chemical components oh inhalable and respirable particulate matter. Int J Environ Res Public Heath 13:592. https://doi.org/10.3390/ijerph13060592
-
Nunes L, Matis J, Catalão J (2016) Biomass combustion system—a review on the concrete and chemical properties of the ashes. Renev Sustain Energy Rev 53:235–242. https://doi.org/ten.1016/j.rser.2015.08.053
-
Padilla-Barrera Z, Torres-Jardón R, Ruiz-Suarez LG, Castro T, Peralta O, Saavedra MI, Masera O, Molina LT, Zavala M (2019) Determination of emission factors for climate forcers and air pollutants from improved wood-burning cookstoves in Mexico. Energy Sustain Develop fifty:61–68. https://doi.org/10.1016/j.esd.2019.02.004
-
PN-EN ISO 17225–1:2014–07 (2014) Solid biofuels—fuel specifications and grades. Part 1: Full general requirements
-
PN-EN ISO 17225–ii:2014–07 (2014) Solid biofuels—fuel specifications and grades. Part two: Wood pellets for non-industrial apply
-
PN-EN ISO 17225–3:2014–07 (2014) Solid biofuels—fuel specifications and grades. Part 3: Wood briquettes for non-industrial use
-
PN-EN ISO 17225–4:2014–07 (2014) Solid biofuels—fuel specifications and grades. Office 4: Wood chips for non-industrial use
-
PN-EN ISO 17225–5:2014–07, (2014) Solid biofuels—fuel specifications and grades. Role 5: Firewood for non-industrial apply
-
PN-EN ISO 18122:2016–06, (2016) Solid biofuels—Determination of ash content
-
PN-EN ISO 18123:2016–01, (2016) Solid biofuels—Determination of volatile matter content
-
PN-EN ISO 18125:2017–07, (2017) Solid biofuel—Determination of calorific value
-
PN-EN ISO 18134–2:2017–03, (2017) Solid biofuels—Determination of wet content—Dryer method. Function 2: Full moisture—Simplified method
-
PN-EN ISO 18135:2017–06, (2017) Solid biofuels—Sampling
-
PN-EN ISO 14780:2017–07, (2017) Solid biofuels—Sample preparation
-
Riddervold IS, Bønløkke JH, Olin A, Grønborg TK, Schlünssen Skogstrand V, Hougaard D, Massling A, Sigsgaard South (2012) Effects of wood smokes particles from wood-burning stoves on the respiratory wellness of atopic humans. Particle Fibre Toxicol ix(12):1–xiii. https://doi.org/10.1186/1743-8977-9-12
-
Ristovski ZD, Wardoyo AY, Morawska L, Jamriska M, Carr S, Johnson 1000 (2010) Biomass called-for influenced particles characteristics in Northern territory Australia Based on airborne measurements. Atmospheric Res 96(1):103–109. https://doi.org/10.1016/j.atmosres.2009.12.002
-
Robinson T, Mohan K, Chilukoti B, Sasmal Southward, Banerjee T, Goud VV (2015) Optimization of dilute acid and hot water pretreatment of different lignocellulosic biomass: comparative written report. Biomass Bioenerg 81:ix–18. https://doi.org/10.1016/j.biombioe.2015.05.006
-
Rybak Due west (2006) Spalanie i współspalanie biopaliw stałych. Wyd. Oficyny Wydawniczej Politechniki Wrocławskiej, Wrocław (in Polish)
-
Sano T, Miura S, Furusawa H, Kaneko S, Yoshida T, Nomura T, Ohara S (2013) Composition of 675 inorganic elements and the leaching behavior of biomass combustion ashes discharged from woods pellet 676 boilers in Japan. J Wood Sci 59:307–320. https://doi.org/x.1007/s10086-013-1337-3
-
Shi R, Li J, Jiang J, Mehmood K, Liu Y, Xu R, Qian W (2016) Characteristics of biomass ashes from different materials and their ameliorative effects on acid soils. J Environ Sci. In Printing, Corrected Proof. https://dx.doi.org/https://doi.org/ten.1016/j.jes.2016.07.015
-
Siergiej P, Jędrak J (2019) Kominki. Czy spalanie drewna jest problemem? Spalanie drewna west kominkach w Polsce i na świecie—wpływ na jakość powietrza i stan zdrowia. Regulacje prawne i polityka antysmogowa w wybranych krajach. Warszawa (Poland) (in Shine)
-
Schmidl C, Marr IL, Caseiro A, Kotianowá P, Berner A, Bauer H, Kasper-Giebl A, Puxbaum H (2007) Chemic characterisation of fine particle emissions from forest stove combustion of cammon forest growing in mid-European Alpine regions. Atmospheric Environ 42:126–141. https://doi.org/x.1016/j.atmosenv.2007.09.028
-
Schober P, Boer C, Schwarte LA (2018) Correlation coefficients: Appropriate use and estimation. Anesthesia Analgesia 126(v):1763–1768. https://doi.org/10.1213/ANE.0000000000002864
-
Supanic K, Oberberger I, Kienzl A, Arich A (2014) Conversion and leaching characteristics of biomass ashes during outdoor storage—results laboratory tests. Biomass Bioenergy 61:211–226. https://doi.org/10.1016/j.biombioe.2013.12.014
-
Symanowicz B, Becher M, Jaremko D, Skwarek K (2018) Possibilities for the use of woods ashes in agriculture. J Ecol Eng 19(3):191–196. https://doi.org/x.12911/22998993/86156
-
Szwalec A, Mundała P, Kedzior R, Telk M, Gawroński P (2016) Zróżnicowanie zawartości Cd, Pb, Zn i Cu w biomasie wykorzystywanej na cele energetyczne (Diverseness of Cd, Pb, Zn and Cu content in the biomass used for free energy purposes). Acta Sci Pol Formatio Circumiectus fifteen(4):343–351. https://doi.org/10.15576/ASP.FC/2016.15.four.343
-
Tao J, Zhang Fifty, Zhang R, Wu Y, Zhang Z, Zhang Y, Tang Y, Cao Y, Zhang Y (2016) Uncertainty assessment of source attribution of PM25 and its h2o-soluble organic carbon content using different biomass burning tracers in positive matrix factorization analysis—a case report in Beijing China. Sci Total Environ 543(vii):326–335. https://doi.org/10.1016/j.scitotenv.2015.eleven.057
-
Thy P, Barfod GH, Cole AM, Dark-brown EL, Jenkins BM, Lesher CE (2017) Trace metal release during wood pyrolysis. Fuel 203:548–556. https://doi.org/10.1016/j.fuel.2017.04.050
-
Trojanowski R, Fthenakis V (2019) Nanoparticle emissions from residential wood combustion: a critical literature review, characterization, and recommendations. Renew Sustain Energy Rev 103:515–528. https://doi.org/10.1016/j.rser.2019.01.007
-
Uliasz-Bocheńczyk A, Mokrzycki Eastward (2018) The elemental limerick of biomass ashes every bit a preliminary assessment of the recovery potential. Mineral Resour Manage 34(four):115–132. https://doi.org/10.24425/122593
-
Vassilev SV, Baxter D, Andersen L, Vassileva CG (2010) An overview of the chemical composition of biomass. Fuel 89:913–933. https://doi.org/10.1016/j.fuel.2009.10.022
-
Vassilev SV, Baxter D, Andersen LK, Vassileva CG, Morgan TJ (2012) An overview of the organic and inorganic phase limerick of biomass. Fuel 94:1–33. https://doi.org/ten.1016/j.fuel.2011.09.030
-
Vassilev S, Baxter D, Andersen L, Vassileva CG (2013) An overview of the limerick and awarding of biomass ash. Part 1. Stage-mineral and chemic limerick and nomenclature. Fuel 105:twoscore–76. https://doi.org/x.1016/j.fuel.2012.09.041
-
Vassilev SV, Vassileva CG, Baxter D (2014) Trace element concentrations and associations in some biomass ashes. Fuel 129:292–313. https://doi.org/x.1016/j.fuel.2014.04.001
-
Vincente East, Alves C (2018) An overview of particulate emissions from residential biomass combustion. Atmospheric Res 199(1):159–185. https://doi.org/10.1016/j.atmosres.2017.08.027
-
Wang S, Miller A, Llamazos Eastward, Fonseca F, Baxter L (2008) Biomass fly ash in concrete: mixture proportioning and mechanical properties. Fuel 87:365–371. https://doi.org/ten.1016/j.fuel.2007.05.026
-
WHO—World Health Arrangement, (2015) World Report on Ageing and Wellness
-
Wiinikka H, Grönberg C, Boman C (2013) Emissions of heavy metals during fixed-bed combustion on half-dozen biomass fuels. Free energy Fuels 27(two):1073–1080. https://doi.org/10.1021/ef3011146
-
Wisz J, Matwiejew A (2005) Biomasa – badania due west laboratorium w aspekcie przydatności exercise energetycznego spalania (Bio-Mass - Laboratory Tests Concerning It Usefulness for Power Combustion). Energetyka 9:631–636
-
Yeboah NNN, Shearer ChR, Burns SE, Kurtis KE (2014) Characterization of biomass and high carbon content coal ash for productive ruse applications. Fuel 116:438–447. https://doi.org/10.1016/j.fuel.2013.08.030
-
Zając G, Szyszlak-Bargłowicz J, Gołębiowski West, Szczepanik M (2018) Chemical characteristic of biomass ashes. Energies eleven:2885. https://doi.org/x.3390/en11112885
Funding
We have not received any funds.
Author information
Authors and Affiliations
Corresponding author
Boosted information
Editorial responsibility: Nour Sh. El-Gendy.
Supplementary data
Rights and permissions
Open Access This article is licensed under a Creative Eatables Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in whatsoever medium or format, equally long every bit you give advisable credit to the original author(s) and the source, provide a link to the Creative Commons licence, and bespeak if changes were made. The images or other tertiary party material in this article are included in the commodity's Creative Commons licence, unless indicated otherwise in a credit line to the cloth. If material is not included in the article's Artistic Commons licence and your intended utilise is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a re-create of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
Near this commodity
Cite this article
Smołka-Danielowska, D., Jabłońska, M. Chemic and mineral composition of ashes from wood biomass combustion in domestic forest-fired furnaces. Int. J. Environ. Sci. Technol. 19, 5359–5372 (2022). https://doi.org/ten.1007/s13762-021-03506-9
-
Received:
-
Revised:
-
Accepted:
-
Published:
-
Issue Date:
-
DOI : https://doi.org/10.1007/s13762-021-03506-9
Keywords
- Chemical composition
- Emission gene
- Fireplace
- Wood ash
Source: https://link.springer.com/article/10.1007/s13762-021-03506-9
Posted by: gardnerthertat.blogspot.com

0 Response to "What Is The Chemical Makeup Of Most Ash And Resin"
Post a Comment